The Hidden Gold in Green Tea: A Scientific Deep Dive into Kaempferol
We often encounter it in our lunch of kale salad or afternoon green tea, yet rarely name it.
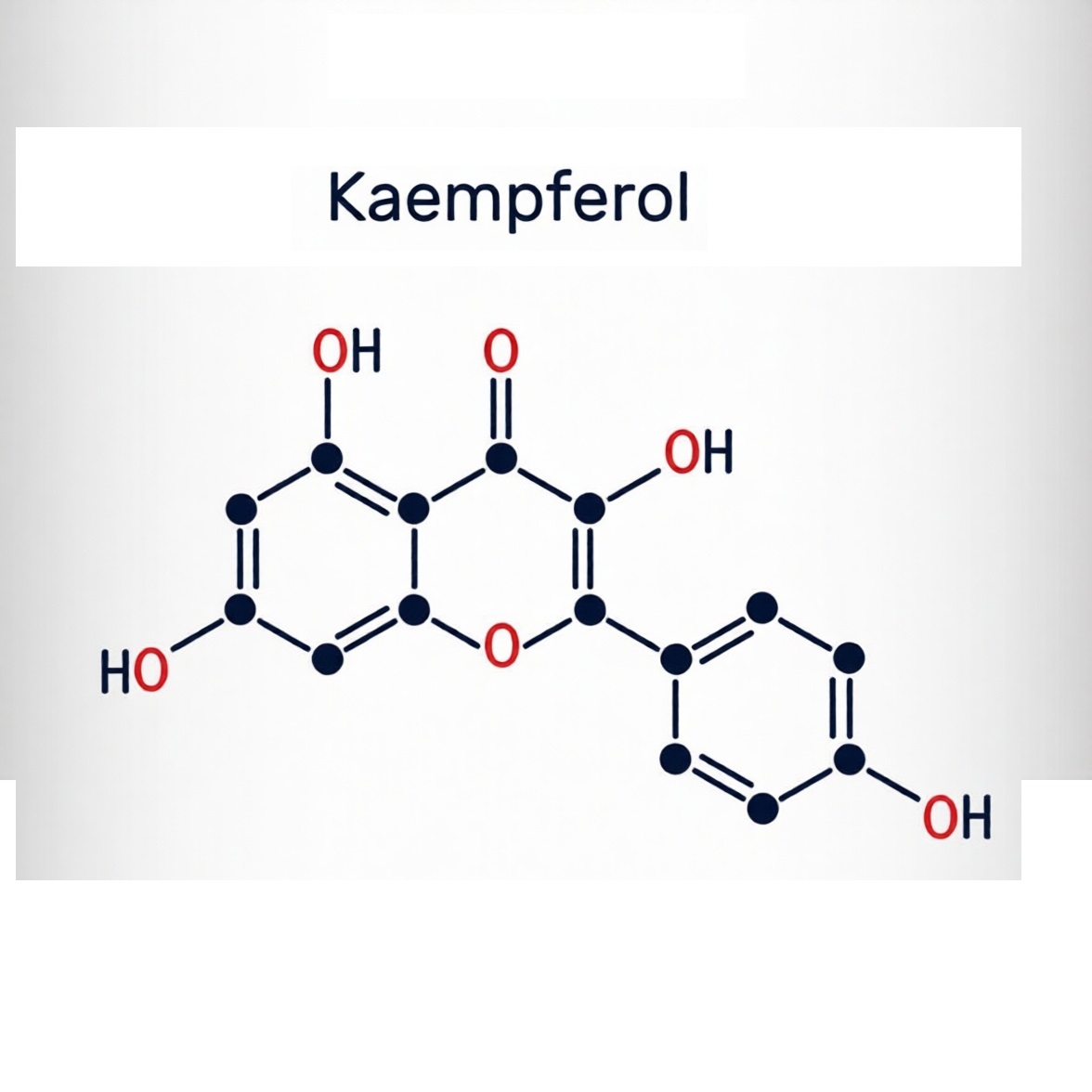
It is Kaempferol, CAS number 520-18-3. To chemists, it is 3,4′,5,7-tetrahydroxyflavone, a yellow crystalline solid with the molecular formula C15H10O6. But to biologists and nutritionists, it is far more than just a plant pigment. It is a silent guardian, a bioactive giant long underestimated due to issues with human absorption.
Today, we won't discuss the vague concept of "superfoods," but rather, as if in a laboratory setting, we'll examine this molecule under a microscope, discussing its past, present, and that fascinating yet perplexing problem of solubility.
History: A Long Journey from the Tea Tree to the Laboratory
The compound's name is actually in honor of the 17th-century German naturalist Engelbert Kaempfer. The very name carries a weight of history—connecting the transmission of knowledge between East and West.
While it is widely found in Moringa, Ginkgo, and Rosmarinus, its most famous "home" is undoubtedly Camellia sinensis (tea tree). Traditional medicine used it for millennia, and it wasn't until the advent of modern chromatography that we were able to truly isolate this yellow powder and confirm its identity. This is not merely the discovery of a component, but the verification of the chemical foundation of ancient wisdom.
The "Big Three" Health Benefits: A Modern Scientific Perspective
When we peel back the marketing jargon, kaempferol's performance at the cellular level is impressive. It's not a panacea, but in these three areas, its performance is textbook-perfect.
1. Cancer Defense Shield (Chemoprevention)
The mechanism here is quite ingenious. Kaempferol appears to possess a degree of "selectivity." Studies show that it can induce apoptosis in specific cancer cell lines (such as colon cancer, breast cancer, and ovarian cancer).
More importantly, it knows when to "stop." While attacking diseased cells, it exhibits extremely low toxicity to normal cells. This isn't magic, but rather a precise strike achieved through the regulation of cellular signaling pathways.
2. Bone Builders (Osteogenic Activity)
This is crucial with the advent of an aging society. Bone health is not just a matter of calcium, but a balancing act between osteoblasts (builders) and osteoclasts (destroyers).
Kaempferol plays a dual regulator role here.
Promoting: It stimulates osteoblast differentiation and mineralization by activating the estrogen receptor (ER) and BMP-2/Smad signaling pathways.
Inhibiting: It simultaneously suppresses RANKL-induced bone resorption. This "boosting" mechanism is highly valuable for the prevention of postmenopausal osteoporosis.
3. Metabolism and Heart Health
It acts like a cellular-level "rust inhibitor." By scavenging free radicals, it reduces oxidative stress in myocardial tissue. Simultaneously, data suggests it contributes to insulin secretion, providing new insights into adjunctive therapy for diabetes management.
Industrial and Analytical Applications: More Than Just a Dietary Supplement
As a researcher with a chemistry background, I must point out that kaempferol's applications in non-biological fields are equally fascinating. Its chemical structure makes it an excellent ligand.
Detective in Analytical Chemistry
In the laboratory, we utilize its sensitivity to metal ions. Due to its specific hydroxyl arrangement, kaempferol can form stable colored complexes with metal ions such as aluminum (Al³⁺), gallium (Ga³⁺), zirconium (Zr⁴⁺), and thorium (Th⁴⁺).
These complexes exhibit a significant spectral shift and produce high-intensity fluorescence. This means it is a natural and highly efficient chromogenic and fluorescent reagent for detecting trace heavy metals in environmental water samples or metallurgical materials.
Food Technology: The Magician of Flavor and Color
If you've ever enjoyed a smooth-tasting plant-based protein drink, you might have something to thank kaempferol. It's a natural bitterness masking agent, effectively reducing the astringency of high-caffeine formulations or plant-based products.
Furthermore, in the wine and juice industries, it acts as a copigmentation agent. It stabilizes anthocyanins through intermolecular interactions, preventing browning. This allows food technicians to maintain the visual appeal of products without using synthetic stabilizers.
The Elephant in the Room: Bioavailability and Solubility
We must face reality. As a substance, kaempferol has a major technical drawback: it is a "stubborn molecule."
Water solubility: <1 mg/mL (virtually insoluble).
Ethanol solubility: ~15 mg/mL.
DMSO solubility: ~20 mg/mL.
This leads to extremely low oral bioavailability. Even if you ingest large amounts, most of it cannot be absorbed in the gastrointestinal tract or is rapidly metabolized by the liver's "first-pass effect." This is currently the biggest pain point in pharmaceutical and health supplement development. Unless liposome encapsulation or nanoparticle formulation technology is used, the effect of simply ingesting powder is often greatly reduced.
Sourcing Guide: Kaempferol (CAS 520-18-3)
For B2B buyers or formulators looking for raw materials, here are some industry insider tips.
1. Purity Pitfalls
The market typically offers 90% and 98% (HPLC) purity.
98%: The preferred choice for pharmaceutical R&D and high-end dietary supplements. You need to confirm that its melting point is between 276–278 °C, a physical indicator of high purity.
90%: May be suitable for some cosmetic applications, but be wary of residual impurities.
2. Price Sensitivity
Why is it so expensive? The extraction process is extremely complex. Isolating this specific flavonoid from a plant matrix requires the use of large amounts of organic solvents and multi-stage purification. The high energy input and low yield mean it won't be a cheap raw material.
3. Compliance Documentation
Don't just look at the price. Always request a Certificate of Analysis (COA) and Material Safety Data Sheet (MSDS).
Safety and Contraindications: A Wise Warning
While it is a natural product, "natural" does not mean "risk-free."
Pregnant women should avoid it: Due to a lack of long-term developmental safety data, and studies suggesting it may cause uterine contractions (especially when consuming large amounts of plants containing kaempferol, such as moringa), pregnant or breastfeeding women should avoid high doses.
Drug Interactions: This is a key clinical concern. Individuals taking **anticoagulants (blood thinners)** are at risk. Kaempferol may enhance the effects of these medications, leading to an increased risk of bruising or internal bleeding.
Mutagenicity Controversy: Early in vitro studies raised concerns about its mutagenicity. However, subsequent in vivo studies have not universally confirmed this, possibly due to its low bioavailability—it is metabolized before it can cause harm. This academic controversy warrants continued attention.
Future Outlook: Nanotechnology and New Frontiers
The story of kaempferol is just beginning.
Current research is focused on solving the "solubility" problem. Through nanoemulsions and polymer micelles, scientists are trying to trick the body's absorption system into allowing this molecule to enter the bloodstream more efficiently.
If this problem is solved, combined with its potential in Alzheimer's disease prevention and synergistic effects with chemotherapy drugs, kaempferol has the potential to leap from a dietary ingredient to a core pillar of next-generation herbal medicines.
Frequently Asked Questions (FAQ)
Q: Which foods contain the most kaempferol?
A: Kale, capers, spinach, broccoli, and green tea are currently known as the best dietary sources.
Q: What is the difference between kaempferol and quercetin?
A: They are structurally similar and often coexist. However, kaempferol is chemically more stable than quercetin and exhibits more unique characteristics in specific bone health pathways.
Q: Why is pure kaempferol powder so expensive?
A: This is due to its complex extraction and purification process. Obtaining a high-purity (98% HPLC) monomer from a large quantity of plant materials requires extremely high technical costs.